 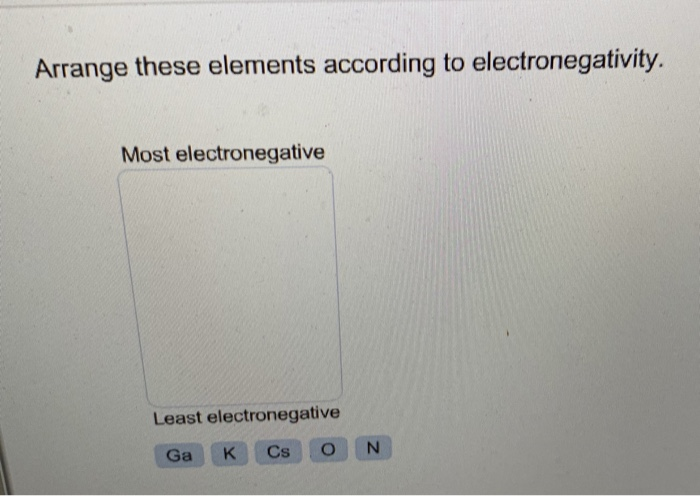
Pauling, The Chemical Bond, Cornell University Press, Ithaca, New York, 1967. Dean (ed), Lange's Handbook of Chemistry (15th Edition), McGraw-Hill, 1999 Section 4 Table 4.5, Electronegativities of the Elements. Pauling, L., The Nature of the Chemical Bond, Third Edition, Cornell University Press, Ithaca, New York, 1960.Boca Raton, Florida, 2003 Section 9, Molecular Structure and Spectroscopy Electronegativity Lide (ed), CRC Handbook of Chemistry and Physics, 84th Edition. Keiter in Inorganic Chemistry : Principles of Structure and Reactivity, 4th edition, HarperCollins, New York, USA, 1993.Īs quoted from these sources in an online version of: David R. 
This is especially problematic for francium, which by relativistic calculations can be shown to be less electronegative than caesium, but for which the only value (0.7) in the literature predates these calculations.  
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
